Coronavirus pandemic is affecting the whole world. The exact antidote for the infection yet not discovered. Several labs across the world are working on an antidote to stop this pandemic. The US also started clinical trials on artificial antibody for treatment of COVID-19. They are facing thousands of the American deaths daily.
China got some positive results
China which is the kind of the origin of coronavirus infection is still trying to find a proper treatment of this virus. Recently China got some positive results when they treated a severely infected coronavirus patient with a single dose of antibodies taken from the blood of recovered patients from COVID-19.
US has begun its clinical trials on an antibody
After this, the US also started clinical trials on treatment. The US is facing thousands of the American deaths daily. The US has started trials on an artificial antibody called gimsilumab. The trial is to evaluate whether it can reduce the risk of death among COVID-19 patients and can help to reduce ARDS, a life-threatening inflammatory lung injury characterized by lack of oxygen to the tissues and stiff lungs.
Gimsilumab
Gimsilumab is an artificially synthesized antibody. This can potentially reduce death risk among COVID-19 patients. It is a man-made antibody. Made by developing a clone of a unique cell that is immune to a particular infection, in an order to manufacture multiple identical immune cells.
The Temple University Hospital researchers have already started treating their first patient using this therapy. Several COVID-19 sufferers experience a hyperactive immune response that leads to lung injury, acute respiratory distress syndrome (ARDS), and ultimately death.
Gimsilumab, monoclonal antibody focused on granulocyte macrophage-colony stimulating factor (GM-CSF), which is believed to be a major factor of lung hyper-inflammation.
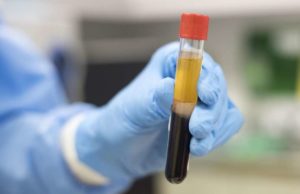
Plasma Therapy
In plasma therapy, an antibody present in the blood of COVID-19 survivors used. A rich amount of blood drawn from COVID-19 survivors. The therapy relies on the survivors’ willingness to donate their blood. For now, it is the most promising treatment against the novel coronavirus. Whereas, gimsilumab can be mass-manufactured in laboratories.
Study
Studies say that COVID-19 patients experience acute respiratory distress syndrome (ARDS). This is a life-threatening inflammatory lung injury and has an overall fatality rate of 41 %. Gimsilumab developed to target the factors which cause such hyper-inflammation of lungs. This is the reason why scientists believe that it will be effective for coronavirus treatment.
Roivant Sciences, a Switzerland-based pharmaceutical company, sponsored the whole trial. They will accept up to 270 confirmed cases of COVID-19 patients who are experiencing ARDS.
In the Temple University Hospital in the U.S. has started intravenous treatment with gimsilumab on at least one patient. The main focus of the new study is to record the incidence of mortality at the end of 43 days. They are also observing the duration of use of ventilator after the therapy. Also the number of days spent in ICUs, or regular hospitalization units.
Previously, gimsilumab tested in several clinical and non-clinical studies, for lung-inflammation, but not in a COVID-19 context.
Dr Mandeep Mehra, Professor of Medicine at Harvard Medical School, told Clinical Trials Arena “The therapy is promising for reducing lung injury and death from the dangerous coronavirus infection. The rapid start of the trial with gimsilumab is impressive, given the pressing need for effective therapies that reduce the morbidity encountered with COVID-19.
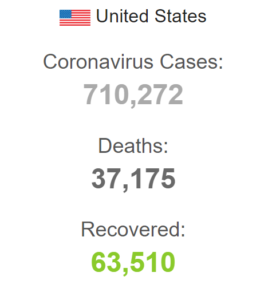
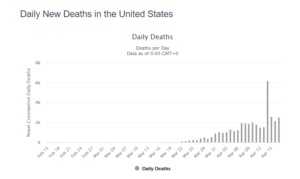
On April 18, the US coronavirus patients reached the count of 710,272 including 37,175 deaths.